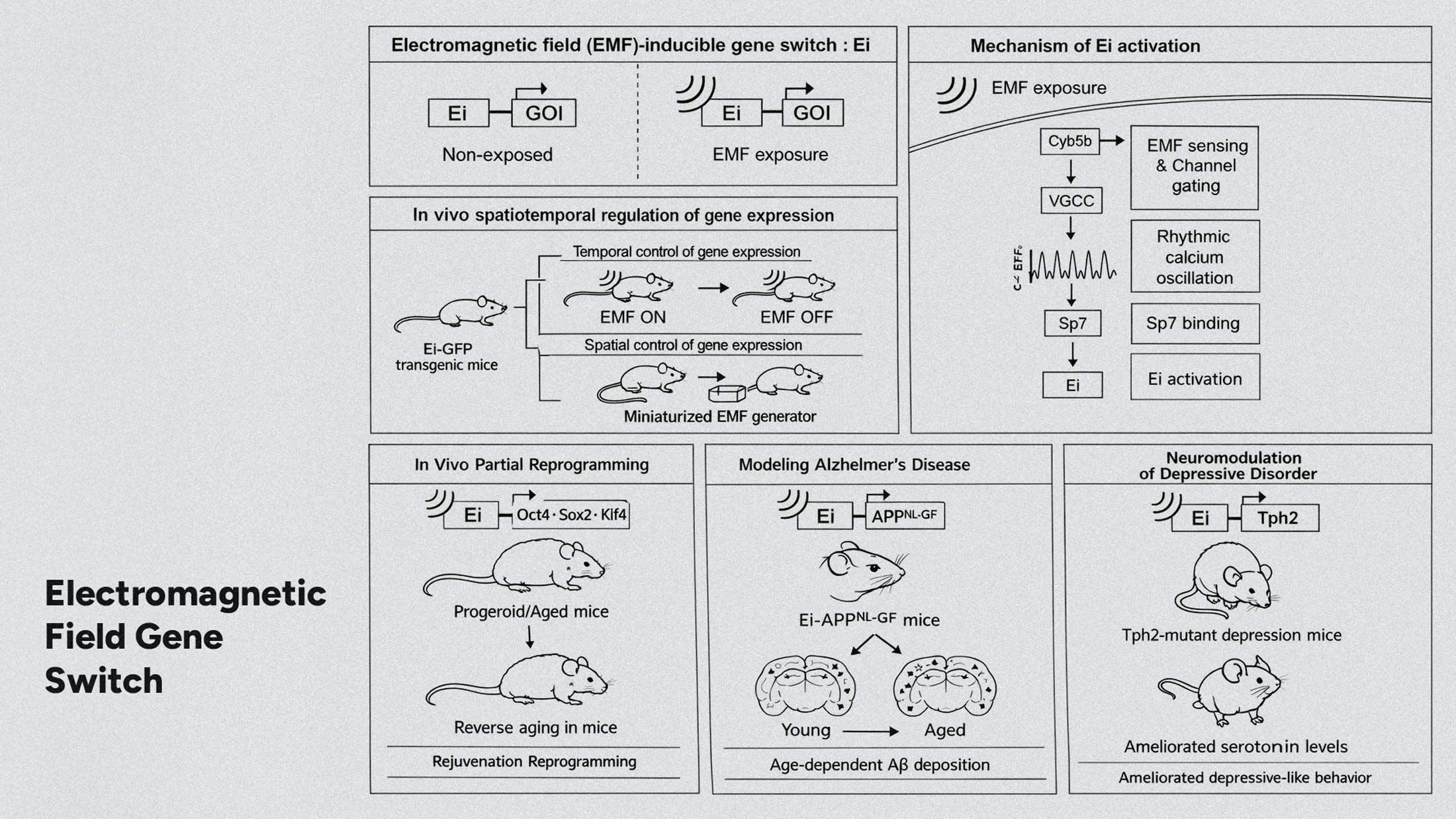
Researchers at Seoul National University have demonstrated something that sounds like science fiction: a gene switch controlled by electromagnetic fields. No drugs. No implants. Just coils generating 60 Hz pulses, turning specific genes on and off in living mice with remarkable precision.
The paper, published in Cell, describes a system that could fundamentally change how we think about genetic medicine. The team activated OSK factors for partial epigenetic reprogramming in aged and progeroid mice, effectively reversing aging markers across multiple tissues and extending lifespan. In a separate application, they used the system to conditionally express mutant amyloid genes, allowing them to isolate Alzheimer's effects from the broader noise of aging.
The Mechanism: CYB5B and Rhythmic Calcium
The research team identified the molecular mechanism through a CRISPR screen. At the center sits CYB5B, a protein that senses rhythmic calcium oscillations generated by the electromagnetic pulses. When CYB5B detects these specific oscillatory patterns, it triggers SP7 to activate target genes.
This distinction matters. Non-specific calcium increases from random stimulation don't produce the same effect. The system responds to rhythm, not just intensity. Think of it as a biological lock that only opens with the right temporal key.
The 60 Hz frequency wasn't arbitrary. It matches standard AC power frequencies in South Korea and much of Asia, suggesting future therapeutic devices could potentially operate using modified versions of common electrical infrastructure.
What This Means for Human Medicine
The implications extend far beyond mouse models. If this mechanism translates to human cells, and early indications suggest the pathway is conserved, we're looking at on-demand gene expression controlled externally.
Consider current gene therapy approaches. Most involve permanent modifications or continuous drug administration. This system offers something different: temporal control. Activate a regenerative pathway when needed, shut it down when the job is complete. The same individual's genome contains instructions for thousands of proteins we rarely express in adulthood. This approach could unlock selective access to that dormant code.
The research also converges with a broader trend. AI systems are increasingly being paired with CRISPR to create what some researchers call CRISPRWARE. The concept treats genetic modification like software development. Design the circuit, test it in silico, deploy it in living systems. Add external control through electromagnetic fields, and you have something approaching programmable biology.
The Remaining Questions
Penetration depth remains unclear. Coils work fine for mice. Human organs sit deeper, surrounded by tissue that may attenuate or distort the fields. The team hasn't published data on larger animals.
Safety over long exposure periods also needs investigation. The 60 Hz fields themselves are generally considered safe, but chronic activation of cellular calcium signaling pathways could introduce risks the short-term mouse studies didn't capture.
Still, the core finding stands. A biological system can be engineered to respond to external electromagnetic signals with genetic specificity. The Seoul team has demonstrated proof of concept. What follows will determine whether this becomes a laboratory curiosity or the foundation of a new therapeutic paradigm.